HEALTHCARE & MEDICAL TECHNOLOGY
AI and Compliance Systems
for Regulated Healthcare .
-
ISO 27001 Certified
-
ISO 22301 Certified
-
GDPR Certified
- Trusted by Nova Leah
HST Solutions delivers AI development and compliance software for medical device companies and healthcare organisations across Ireland, the UK, Europe, and the US, including NLP automation, regulatory document processing, and FDA/CE aligned systems. ISO 27001 certified.
Why Healthcare and Medtech Companies Bring Us In
- Regulatory compliance is manual and slow
- AI prototypes don't reach production
- Hiring engineers with healthcare experience is brutal
- Security requirements are non-negotiable
You don’t need another AI demo. You need compliant systems that
automate regulatory burden and ship through FDA/CE approval.
Who brings in HST for healthcare
Medical device companies automating compliance MDS2, cybersecurity questionnaires, regulatory submissions
Healthcare AI companies moving models to production from research notebooks to deployed systems
Digital health startups scaling platforms with the architecture to pass regulatory scrutiny
Clinical organisations modernising data infrastructure while maintaining HIPAA/GDPR compliance
Medtech firms preparing for FDA/CE submission who need documented, auditable software development
If that sounds familiar, we've solved it before.

What is Medical Device Software?
Medical device software encompasses software that is either a medical device itself (Software as a Medical Device / SaMD) or software that is part of a medical device. This includes diagnostic algorithms, clinical decision support, patient monitoring systems, and device management platforms.
Medical device software development is governed by strict regulations including FDA 21 CFR Part 820, EU MDR, IEC 62304 (software lifecycle), and ISO 13485 (quality management). The global medical device software market is projected to reach $103 billion by 2031, growing at 12.3% CAGR.
HST provides senior engineers who build healthcare and medical device software with the documentation, security, and quality standards required for regulatory approval
AI & Machine Learning
- Natural Language Processing for document automation
- Clinical decision support algorithms
- Diagnostic prediction models
- Medical image analysis
- Anomaly detection and alerting
Regulatory Compliance Automation
- MDS2/MDS+ questionnaire automation
- Cybersecurity compliance (FDA premarket guidance)
- Document similarity and matching
- Regulatory submission preparation
- Audit trail and evidence generation
Data Platforms
- Healthcare data lakes (HIPAA/GDPR compliant)
- Clinical data integration
- Real-world evidence platforms
- Patient registry systems
- Interoperability (HL7 FHIR, DICOM)
Custom Software Development
- Clinical workflow applications
- Patient portals and engagement tools
- Device management platforms
- Healthcare provider tools
- Integration with EHR/EMR systems
Case Study

Nova Leah — AI/NLP for Medical Device Cybersecurity
Nova Leah helps medical device manufacturers assess and manage cybersecurity risks. Their platform automates compliance with FDA premarket cybersecurity guidance and supports MDS2 (Manufacturer Disclosure Statement for Medical Device Security) questionnaire completion.
- Challenge
- AI/NLP system for automated questionnaire completion
- Fuzzy matching and semantic similarity (distilRoBERTa) for answer suggestions
- 9 microservices architecture on Azure Functions
- Cosmos DB for document storage and retrieval
- Confidence scoring and human in the loop review workflow
- What We Built
- Migrated core holdings and risk data to AWS S3/Iceberg data lake
- Implemented Glue, DMS, and CDC for automated data pipelines
- Deployed Athena for analyst querying with Lake Formation governance
- Replaced fragile SSIS packages with monitored, tested Glue jobs
- Created comprehensive runbooks for operations team
- Technology
- Python | Azure Functions | Cosmos DB | distilRoBERTa | Semantic similarity | Microservices
- Result
- Answer time reduced from hours to minutes. Regulatory experts now review AI suggestions rather than starting from scratch. Consistency improved across product lines. Platform scaled to support multiple medical device manufacturers.
TyreCheck — Multi-Agent RAG System
TyreCheck required an intelligent system to route technical queries to appropriate knowledge sources and generate accurate responses.
- What We Built
- Multi agent RAG (Retrieval Augmented Generation) architecture
- Semantic similarity agent for document matching
- Query generation agent for database lookups
- Fallback LLM for edge cases
- Pipeline orchestration for agent coordination
- Technology
- Cosmos DB | RAG | Semantic similarity | LLM | Multi-agent architecture
- Result
- Reduced incorrect responses, enabled 24/7 scalable technical intelligence, improved query resolution time
Why healthcare companies choose HST over alternatives
|
Big 4 / Consultancies | Offshore Teams |
| |
|---|---|---|---|
| Healthcare domain | Generic consultants | None | Built for Nova Leah, understand regulations |
| AI to production | POCs and decks | Model-only | Full deployment with compliance docs |
| Security posture | Variable | Questionable | ISO 27001 & 22301, HIPAA-ready |
| Documentation | Change requests | Minimal | Audit ready from day one |
| Speed | 8-12 weeks | Communication gaps | 7-10 days |
We've shipped AI systems through healthcare compliance review.
We know what auditors look for.
Ireland
Europe's Medtech Hub
Ireland is Europe’s largest medtech employer per capita:
50,000+
€570 million VC raised
18 of top 25
Medtronic, Boston Scientific, Stryker, Abbott
200+ digital health businesses
Ireland’s medtech cluster benefits from a skilled workforce, favourable regulatory environment, and proximity to European markets. HST operates in this ecosystem with deep healthcare technology experience.
Regulatory & Compliance Expertise
| Regulation | What We Deliver |
|---|---|
| FDA 21 CFR Part 820 | Quality system compliant development processes |
| EU MDR | Documentation and technical file support |
| IEC 62304 | Software lifecycle documentation |
| ISO 13485 | Quality management aligned development |
| HIPAA | Compliant architecture, access controls, encryption |
| GDPR | Patient data governance, consent management |
| FDA Cybersecurity Guidance | Premarket submission support |
Our ISO 27001 and ISO 22301 certifications demonstrate security and business continuity standards aligned with healthcare requirements. We build with compliance in mind from day one.
Services for Healthcare
| Service | Application in Healthcare |
|---|---|
| AI/ML Engineering |
|
| Data Engineering |
|
| Custom Software Development |
|
| Cloud Security & DevSecOps |
|
| DevOps & CI/CD |
|
Technology Stack

























We build for healthcare compliance. Every design decision considers regulatory requirements.
Pricing
Clear, guarded, no hidden overages
Precision Pod
€5–6k/month
Single seat
- 1 Senior AI/Software Engineer
- PM included (up to 6–8h/month)
- Architecture reviews (up to 2h/week)
- DevOps/QA assist
- 3 month minimum, then 30 day notice
Pair Pod
€10–11k/month
Two engineers
- 2 Senior Engineers (e.g., AI + Backend)
- PM included (up to 10h/month)
- Architecture reviews (up to 4h/week)
Mini-Team
€15–16k/month
Three engineers
- 3 Engineers (e.g., AI + Backend + DevOps)
- PM included (up to 15h/month)
- Architecture reviews (up to 6h/week)
- Swap guarantee in first 2 weeks.
Start in 7–10 business days.
Give us 20 minutes. We'll show you how we've built AI systems for medical device companies.
Frequently asked questions
What is Software as a Medical Device (SaMD)?
SaMD is software intended to be used for medical purposes without being part of a hardware medical device. Examples include diagnostic apps, clinical decision support software, and treatment planning tools. SaMD requires regulatory approval (FDA clearance/CE marking) and must be developed under quality management systems like ISO 13485.
Do you develop FDA-regulated medical device software?
We develop software for medical device companies that goes through FDA and CE regulatory processes. We build with IEC 62304 lifecycle documentation, maintain audit trails, and create the technical documentation required for regulatory submissions. Our ISO 27001 certification supports the security requirements regulators expect.
Can you build HIPAA-compliant healthcare applications?
Yes. We architect healthcare systems with HIPAA compliance from the start encryption at rest and in transit, access controls, audit logging, and data governance. Our cloud deployments use HIPAA eligible services on AWS and Azure with appropriate BAA coverage.
What AI/ML capabilities do you have for healthcare?
We build production AI systems including NLP for clinical document processing, computer vision for medical imaging, predictive models for clinical decision support, and classification systems for diagnostics. We deploy these with proper validation, monitoring, and the documentation healthcare regulators require.
How do you handle healthcare data security?
Security is foundational, not an add on. We’re ISO 27001 certified. We architect with defence in depth network segmentation, encryption, access controls, audit logging, vulnerability scanning. For healthcare specifically, we align with HIPAA Security Rule requirements and implement technical safeguards appropriate to the data sensitivity.
How fast can you start?
7–10 business days from signed agreement to engineer embedded in your team.
Find The Perfect Solutions For Your Project
Managed Team
Your product, our dedicated team. From concept to conception, we handle it all.
Staff Augmentation
Need extra hands? Our experts seamlessly join your team, providing the skills you need, when you need them.
Fixed Cost
One Team, One Dream
Build Trust with Every Interaction
Improve Everything
Own It
Obsessed: Over Results
Proven Excellence
Partners in Precision
Who Are We ?
Creativity, Efficiency, & Advanced AI
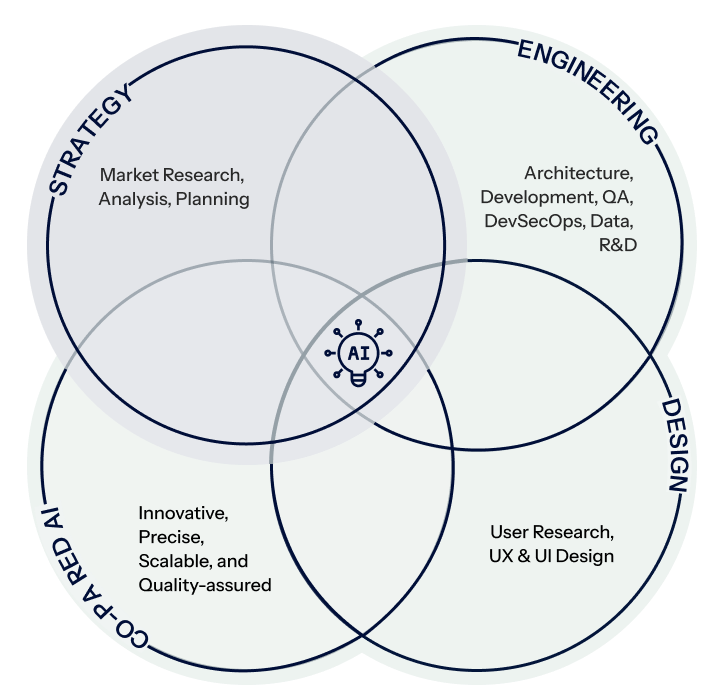
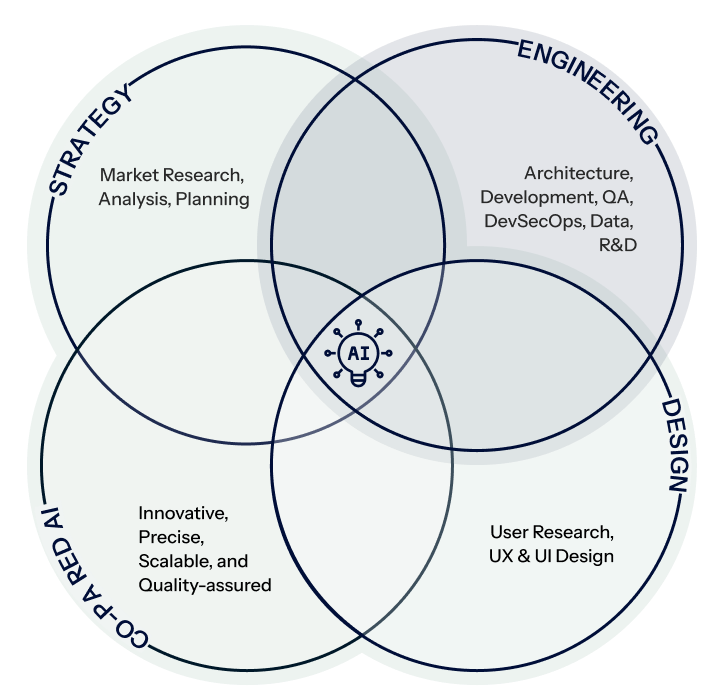
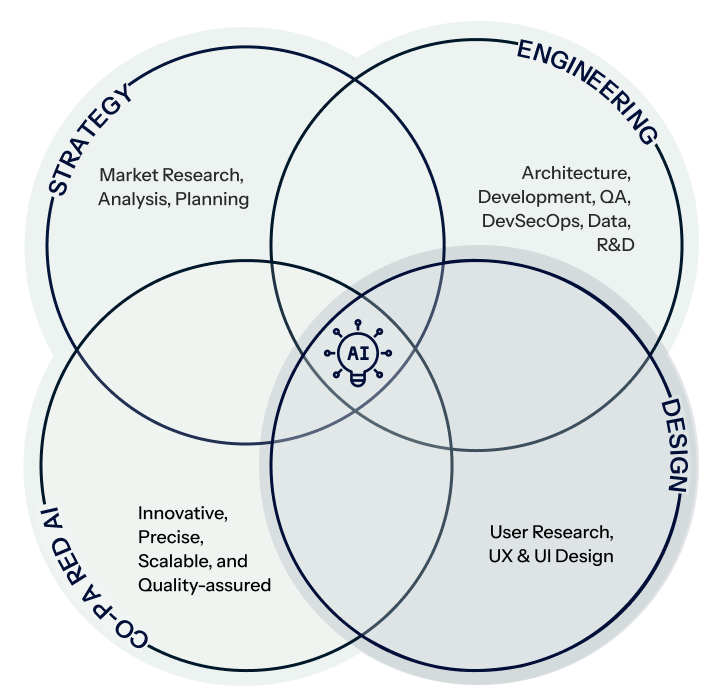
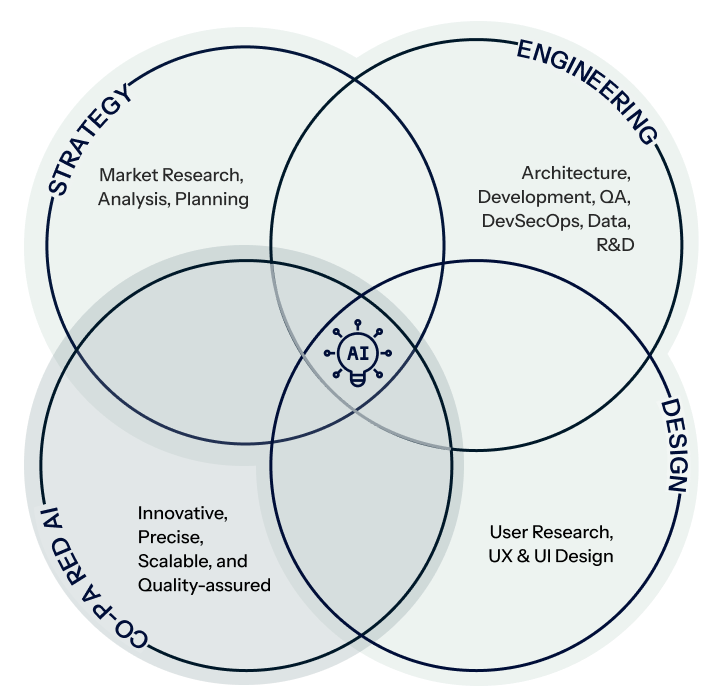
Strategy
Engineering
Design
Co-paired AI

Strategy

Engineering

Design

Co-paired AI
Contact Us
Tell us about your custom software project
Let our team, be your team
Get a technical conversation about your project — not a slide deck. Whether you need AI integration, a software engineering team, or a data platform, we’ll tell you honestly if we’re the right fit.



